It is obvious, it will be very useful in analysis of both thermodynamic cycles used in power engineering, i.e. in isentropic process, the enthalpy change equals the flow process work done on or by the system: At constant pressure, the enthalpy change equals the energy transferred from the environment through heating:Īt constant entropy, i.e. As can be seen, this form of the law simplifies the description of energy transfer. This work, Vdp, is used for open flow systems like a turbine or a pump in which there is a “dp”, i.e. In this equation the term Vdp is a flow process work. Since H = U + pV, therefore dH = dU + pdV + Vdp and we substitute dU = dH – pdV – Vdp into the classical form of the law: A negative value indicates that work is done on the system by its surroundings. A positive value for work indicates that work is done by the system on its surroundings. 
In this case the final state is the same as the initial state, but the total work done by the system is not zero. Moreover, we can take the system through a series of states forming a closed loop, such i ⇒ f ⇒ i. 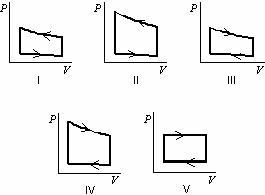
The second process shows, that work is greater and that depends on path of the process. Work W is positive because the system’s volume increases. The blue area represents the pΔV Work done by a system as it goes from an initial state i to a final state f. As can be seen from the picture (p-V diagram), work is path dependent variable. Q and W are path dependent, whereas ΔE int is path independent. One of the most important conclusions is that: When they are all equilibrium states, the path can be plotted on a pV-diagram. There are always infinitely many different possibilities for these intermediate states. When a thermodynamic system changes from an initial state to a final state, it passes through a series of intermediate states. The work done by the system depends not only on the initial and final states, but also on the intermediate states-that is, on the path. The first law of thermodynamics and the work can then be expressed as: To calculate such processes, we would need to know how pressure varies with volume for the actual process by which the system changes from state i to state f. the work done is:ĭuring the volume change, the pressure and temperature may also change. Therefore the resulting volume is 2 m 3 x 1.67 = 3.34 m 3 and ∆V = 3.34 m 3 – 2 m 3 = 1.34 m 3. Since at this enthalpy the steam have density of 1.31 kg/m 3, it is obvious that it has expanded by about 2.2/1.31 = 1.67 (+67%). When we use simply Q = H 2 − H 1, then the resulting enthalpy of steam will be:įrom steam tables, such superheated steam (15812/4.4 = 3593 kJ/kg) will have a temperature of 828 K (555☌). Since at this condition the steam has density of 2.2 kg/m 3, then we know there is about 4.4 kg of steam in the piston at enthalpy of 2912 kJ/kg x 4.4 kg = 12812 kJ. Using steam tables we know, that the specific enthalpy of such steam (500 kPa 500 K) is about 2912 kJ/kg. Calculate the final temperature, if 3000 kJ of heat is added. 
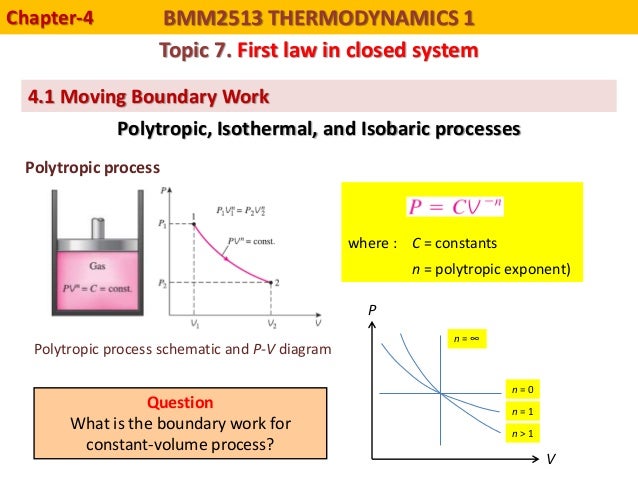
This is what happens when steam, or gas contained in a piston-cylinder device expands against the piston and forces the piston to move.Ĭonsider a frictionless piston that is used to provide a constant pressure of 500 kPa in a cylinder containing steam ( superheated steam) of a volume of 2 m 3 at 500 K.Ĭalculate the final temperature, if 3000 kJ of heat is added. It is used for calculating piston displacement work in a closed system. Boundary work (or pΔV Work) occurs when the volume V of a system changes. Boundary work occurs because the mass of the substance contained within the system boundary causes a force, the pressure times the surface area, to act on the boundary surface and make it move. The pΔV Work is equal to the area under the process curve plotted on the pressure-volume diagram. Pressure-volume work (or pΔV Work) occurs when the volume V of a system changes. PΔV Work is equal to the area under the process curve plotted on the pressure-volume diagram. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
